As India continues its battle against the second wave of Covid-19, it launched its first batch of indigenous anti-COVID oral drug 2-deoxy-D-glucose (2-DG) developed by the Institute of Nuclear Medicine and Allied Sciences (INMAS), a lab of the Defence Research and Development Organisation (DRDO) on Monday.
The drug, called 2-deoxy-D-glucose or 2-DG, was developed by a DRDO lab in collaboration with the Hyderabad-based pharma giant, Dr Reddy’s Laboratories. The Drugs Controller General of India (DCGI), the country’s top drug regulator, has approved the medicine for emergency use as an adjunct therapy in Covid patients battling moderate to severe symptoms of the infection.
The first and the second batch of 2-DG will be used in a limited manner. It will be used in AIIMS, Armed Forces Hospitals, DRDO hospitals and any other need which arises. From June 2021 onwards it will be made available to all hospitals.
The first & second batch (of anti-COVID drug 2-DG) will be used in a limited manner. It will be used in AIIMS, Armed Forces Hospitals, DRDO hospitals & any other need which arises. From June onwards it will be made available to all hospitals: Dr G Satheesh Reddy, Chairman, DRDO pic.twitter.com/VCK2GGVCFp
— ANI (@ANI) May 17, 2021
Defence Minister Rajnath Singh and Union Health Minister Dr Harsh Vardhan released the first batch of 10,000 doses of the medicine at DRDO headquarters today. These doses have been distributed to some hospitals in the national capital.
“Not just for India but I hope it serves in the fight against COVID, globally in the coming days. I thank and congratulate DRDO and its scientists. We’ve seen under PM Modi leadership DRDO has played an important role in the fight against coronavirus.” Union Health Minister Harsh Vardhan said.
Moreover, while launching the drug, Defence Minister Rajnath Singh also called it a new ray of hope. “Developed by DRDO and DRL, 2-deoxy-D-glucose (2-DG) will prove effective against Covid-19. This is a great example of the scientific prowess of our country. I congratulate DRDO and all the institutions involved in the R&D of this drug, on my behalf,” said Singh.
DRDO’s 2-DG drug
On May 9 2021, DRDO Chief Mr G Satheesh Reddy had announced that the DRDO Covid medicine is approved for emergency use and will be available by May 11, 2021.
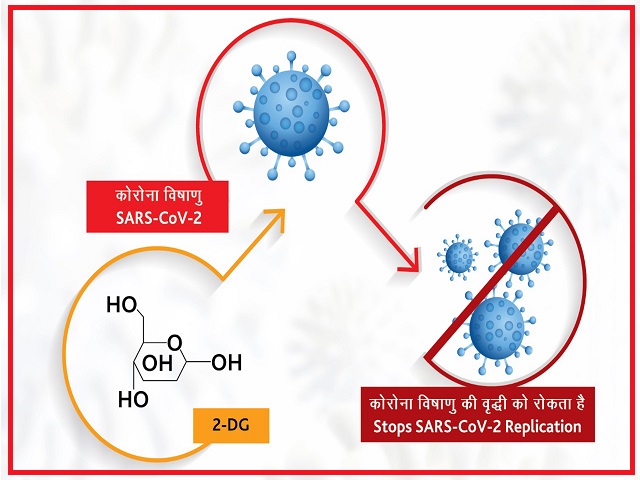
2-deoxy-D-glucose or 2-DG is an oral anti-Covid-19 drug. The drug is expected to help hospitalised Covid-19 patients and reduce their supplemental oxygen dependence.
Explaining what went behind the development of India’s first indigenous anti-COVID oral drug, DRDO Project Director and Scientist of 2-DG, Dr Sudhir Chandana had said earlier that when India was grappling with the first wave of the pandemic in April 2020, the institute discovered that 2-DG halts the spread of COVID-19 inside the body cells.
“After the findings, we asked the DCGI for permission to conduct clinical trials. In May 2020, we got permission for the clinical trials”, Chandana said.
According to reports, a total of 110 patients were part of the Phase-II clinical trials of DRDO’s 2-DG drug. “By the end of October 2020, we had completed the second phase of trials, and the results were very good. Using standard care, 2 DG will be more beneficial for the Covid-19 patients,” Chandana added.
The results of 2-DG phase II trials showed that in terms of improvement in the vitals of COVID-19 symptomatic patients there was a difference of 2.5 days compared to Standard of Care (SoC).
Meanwhile, the DRDO statement had confirmed earlier that the approval for Phase – III clinical trials of 2-DG was granted in November 2020. These were conducted in 27 COVID-19 hospitals spread across several states. “Clinical trial results have shown that this molecule helps in faster recovery of hospitalised patients and reduces supplemental oxygen dependence,” the official DRDO statement had said.
How 2-DG works and controls COVID-19, reducing dependence on oxygen?
According to DRDO officials, the 2-DG drug, like glucose, spreads through the body and reaches the virus-infected cells and prevents virus growth by stopping viral synthesis and destroying the protein’s energy production.
The drug also works on virus infection spread into the lungs which help us to decrease the infected patient’s dependency on oxygen.
Production and consumption and the expected price of the drug
Since 2-DG is a generic molecule and analogue of glucose it can be easily manufactured in India and made available in plenty to the citizens.
The anti-Covid drug 2-DG has been developed in powder form and is ingested orally in patients by dissolving it in water.
According to sources, the price of the drug may range from anything between Rs 500 to Rs 600. However, the government may also grant subsidies making it further affordable for patients from every stratum of society.
Which variants of Covid-19 virus will 2-DG work on
According to DRDO, clinical trials of the medicine has proved that the drug can arrest any variant of the virus that multiplies. Irrespective of the variant it will need food and thus will get trapped by the drug, informed the officials.
DRDO calls the 2-DG drug a game-changer
DRDO’s anti-covid drug is being seen as a boon, especially at a time when the devastating second wave of the infection has led to acute oxygen shortage in the country. The officials believe that the drug will be a game-changer as it will help in faster recovery of patients who are hospitalised especially reducing the supplemental oxygen dependence in such patients.
“In 2-DG arm, a significantly higher proportion of patients improved symptomatically and became free from supplemental oxygen dependence (42% vs 31%) by Day-3 in comparison to SoC, indicating an early relief from oxygen therapy/dependence. A similar trend was observed in patients aged more than 65 years,” the DRDO said.
A higher proportion of patients treated with 2-DG showed RT-PCR negative conversion in COVID patients. In the efficacy trends, the patients treated with 2-DG showed faster symptomatic cure than Standard of Care (SoC) on various endpoints, the DRDO said.