Recently, after massive criticism and NSA-level talks, the United States administration has announced that they would allow the export of crucial raw material required to produce the vaccine in India. Amidst all the lobbying, political, diplomatic and ‘intellectual’ talks over raw material, for the layman, and sometimes for “experts”, it gets difficult to understand why India needs Covid-19 vaccine ingredients from the US.
What was so crucial that India cannot produce or procure from other country? Was there any patent issue or some IP issue, or it was just like the PPE kits that we used to import till the time there was no way to import it anymore? To make things clear, the situation in the case of vaccines is completely different from PPE kits.
Vaccine production requires 9,000+ materials from 300 suppliers from 30 nations
According to World Trade Organization’s (WTO) information note about issues with trade impact titled “Developing And Delivering Covid-19 Vaccines Around The World”, vaccine manufacturing across the world is supported by complex upstream raw material and component value chains. Any typical vaccine manufacturing plant will use approx 9,000 different materials that it source from some 300 suppliers across approx 30 different countries. The estimate is based on the data provided by the International Federation of Pharmaceutical Manufacturers and Associations.
In the first six months of 2020, the demand for critical raw materials for vaccine production, including nucleic acids, amino acid phenols, acyclic amides, lecithins, and sterols, grew by 49% and reached a value close to USD 15.5 billion. The United States is the largest producer of some of the critical raw materials, especially bioreactor bags, filters, and more.
Adar Poonawalla has been voicing concerns over raw materials supply for weeks. The SII had millions of doses of Covishield ready when the approvals came, an achievement that needed an investment of millions of dollars to ramp up productions and a lot of risks. Unless the US embargo is lifted, the SII’s attempts to stockpile Novavax vaccine doses may be impacted, Poonawalla had said.
As per reports SII currently manufactures over 170 million doses of both Covishield and Covovax.
The raw materials that Indian manufacturers require
Based on the statements made by Adar Poonawalla of SII, Dr. Krishna Ella of Bharat Biotech, Mahima Datla of Biological E, and US officials, the raw materials that Indian manufacturers require include Bioreactor bags, filters, cell culture media, microcarriers, Lipid Nanoparticles, and much more.
Bioreactor bags: These are disposable plastic bags that are used in the process of artificially growing a large quantity of cells required for virus cultivation for the vaccine. These bags are also used to mix various ingredients. These bags are necessary for production only if the company is not using a static stainless-steel bioreactor, such as in the case of SII. Such bags are used in the development of inactivated, viral vector, protein subunit, and mRNA based vaccines. These are produced majorly by American multinational company Baxter Healthcare, Massachusetts-headquartered ThermoFisher and Cytiva.
Filters: These filters can be made out of different materials depending on the requirement. They allow clarification, purification and sterilization of the vaccine after the virus is cultivated. Such filters are available in different pore-sizes. They are used in the production of inactivated, viral vector, protein subunit, and mRNA based vaccines. The filters are majorly supplied by companies like Pall Life Sciences (New York) and Merck Millipore, owned by Germany’s Merck but headquartered in Massachusetts.
Cell culture media: It is a gel or liquid-based material that helps in the growth of the cells in the labs. It provides nutrients and compounds to accelerate cell growth. It is used in the production of inactivated, viral vector, protein subunit, and mRNA based vaccines. Cytiva-owned HyClone and Merck Millipore are significant suppliers from the US, but Germany’s CellGenix, India’s HiMedia and Switzerland’s Lonza Group AG also make these.
Lipid Nanoparticles: Used specifically in mRNA-based vaccines, these nanoparticles are used to encapsulate the drug substance for its delivery. US vaccine manufacturers like Pfizer-BioNTech and Moderna require this material for the mRNA-based vaccine. Recently, an Indian company VAV Lifesciences has cracked a major deal with American contract development and manufacturing firm for phospholipids which will be used in the production of vaccines of Pfizer-BioNTech and Moderna.
Microcarriers or Microcarrier beads: These are tiny spheres made out of non-toxic material. These are mainly used in the production of inactivated, viral vector, and protein subunit-based vaccines. They are used to grow protein-producing or virus-generating cells. American companies like Pennsylvania’s VWR International and Cytiva, as well as Germany’s Sartorius, make these microcarriers.
Dr. Gagandeep Kang, the vice-chair of the Coalition for Epidemic Preparedness Innovations (CEPI) and a professor at the Christian Medical College (CMC) in Vellore, was quoted in an Indian Express report saying that while most of the equipment manufacturing is done in regions like Europe, US companies have a stronghold on the supplies of plastics and the bulk of reagents used in a laboratory.
Understanding US embargo
Biden’s administration has used the US defence Production Act, 1950, to block the export of critical raw material in January. The law was first used in 1950 during the Korean war to ensure supplies and equipment. Since then, the scope of the law has been extended beyond the military to cover natural hazards, terrorist attacks and national emergencies. The aim of Biden’s administration was to ensure the domestic businesses and corporations fulfil their federal contracts.
The law was also used by former President Donald Trump to increase the production of ventilators and limit the export of medical supplies. It also aided in increasing supply for ramping up the production of Pfizer and BioNTech vaccines. In the case of the Biden administration, the act was invoked to ensure the supply of products to ensure 24×7 manufacturing of the Johnson & Johnson vaccine.
In March 2021, Financial Times quoted Mahima Datla, chief executive of pharmaceutical company Biological E, the company that is producing J&J vaccine and a protein subunit vaccine with Houston’s Baylor College of Medicine. Datla said the US suppliers had shown an inability to fulfil orders to global clients due to the Defense Production Act invoked by the administration.
How the US Embargo is impacting supplies
As per reports, the restrictions imposed by the United States have put pressure on the major suppliers across the world. If the restrictions continue further, there will be a fight for the resources. If the companies like SII are expected to produce over a billion doses every year, they need unrestricted supplies of the raw material which they are unable to procure.
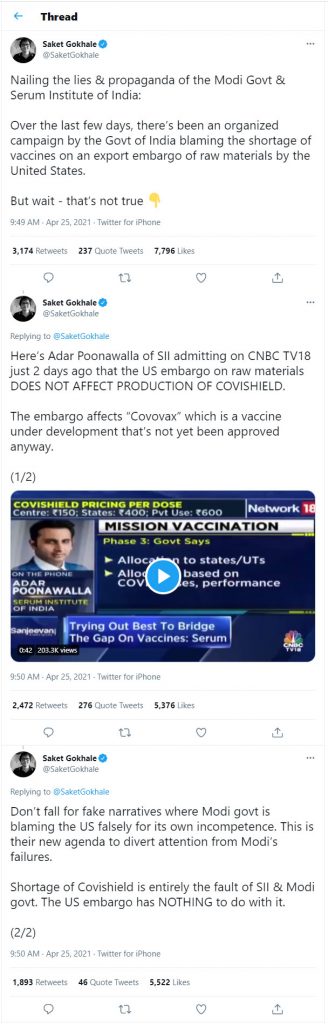
There are some misleading claims that Adar Poonawala had said embargo does not affect the Covidshield production. It only affects the production of Covovax, which is awaiting approval from the government.
It is true that SII has ‘managed’ to keep producing Covidshield irrespective of the embargo. However, it has to be noted that SII has been making Covidshield much before the approval to ensure there is enough stock available once the approval was done. The same goes for the Covovax vaccine. Though the approval is pending, it is expected that the company will get approval from the government in a matter of few weeks. At that time, to ensure there is enough stock of the vaccine available, the company has to ramp up the production. It cannot wait for the approval to come and then work on manufacturing the vaccine. It is a simple matter of common sense that the alleged ‘experts of all fields’ fail to comprehend.
Poonawala reportedly said in one of his statements that the restrictions imposed by the US government have halved its capacity to stockpile Covovax, which would have added to the problem in the future. He also categorically said that though the shortage of raw material has not impacted the production of Covidshield so far, it may impact “the scaling up of Covishield’s future capacity”.
He said, “This is one thing that would need some discussion with the Biden administration to explain to them there’s enough to go around.” He further added, “We’re talking about having free global access to vaccines but if we can’t get the raw materials out of the US — that’s going to be a serious limiting factor.” Poonawala and other manufacturers in India have been voicing their concerns for the last three months since the ban was imposed.
It is notable here that Adar Poonawalla’s SII is the world’s largest vaccine manufacturer. It has been signed up by GAVI’s Covax alliance to supply over 2 billion doses of Covishield and Covovax.
SII is not the only company impacted by the decision
The US Act is not only impacting SII’s ability to produce the vaccine but also other companies like Bharat Biotech and Biological E. Dr Krishna Ella, the chairman and managing director of the Hyderabad-based Bharat Biotech that makes Covaxin had said in March that the restrictions had impacted the supply logistics for vaccine makers.
The US Act seems to be impacting the ability of other Indian companies to make their Covid-19 vaccines in India. For instance, Dr Krishna Ella, the chairman and managing director of the Hyderabad-based Bharat Biotech that makes Covaxin, said late in March that the restrictions by the US “on some of the materials” have impacted supply logistics for vaccine makers. “You see the supply logistics, where the US has put a restriction on some of the materials (for making vaccines). They cannot be exported to other countries. Actually, one of the raw materials we need to get, we are not able to get it from the US and Sweden,” said Dr Ella.
Datla, while talking to Financial Times, had said that it has become extremely difficult for the vaccine manufacturers to not only produce Covid-19 vaccine but routine vaccines as well. Her company is expected to make a billion doses of the J&J vaccine and scale up production of its recombinant protein vaccine with Baylor College to a billion doses over an unspecified time period. She added that it was not clear how the restriction would impact the production in future as the company was testing the recombinant protein vaccine in India.
WHO had also expressed concerns over export embargo
SII’s Adar Poonawalla has been repeatedly making appeals at international forums for nations to not impose export restrictions. Speaking at an World Bank event in March 5, Poonawalla had stated that US embargo over key raw materials will cause severe bottlenecks for vaccine production.
If we are talking about building capacity all over the world, the sharing of these critical raw materials is going to become a critical limiting factor. Nobody has been able to address this so far.”
Soumya Swaminathan, the chief scientist of the WHO, had also expressed concern over the availability of raw materials. “There is a shortage of raw materials, of products that you need for the manufacturing of vaccines. This is where you need global agreement and coordination not to do export bans.”
There are other suppliers, but the situation is complicated
Though there are other companies in the world that can provide the raw materials, but, at this time, due to the surge in demand, logistics, and regulatory issues, it is difficult to hunt for another suppliers. Moreover, unlike other medical devices, when materials for the vaccine are sourced from another supplier, the drug controller has to be informed. That means if any of vaccine manufacturer wants to change the suppliers, they would need permission from the drug controller that is a lengthy process. Even the standard manufacturing protocols will need changes in certain cases, further causing regulatory concerns, not to mention the impact on the production process and quantities. Thus, getting permissions in time for a change of suppliers is not feasible, especially at the time when India is facing an unexpected surge due to the second wave of the Covid-19.